 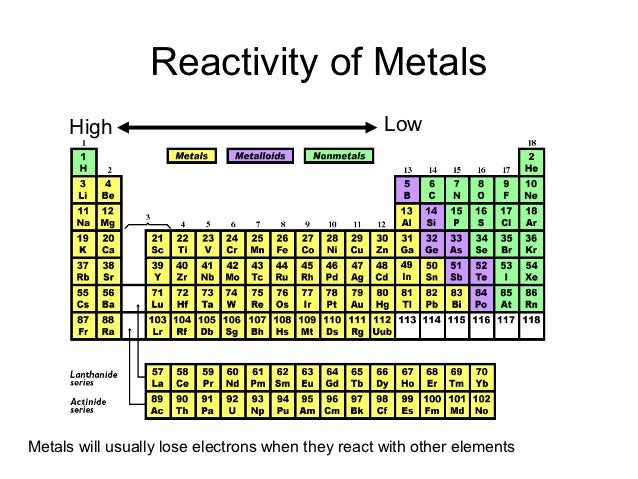
It increases on moving from left to right as more energy is released. 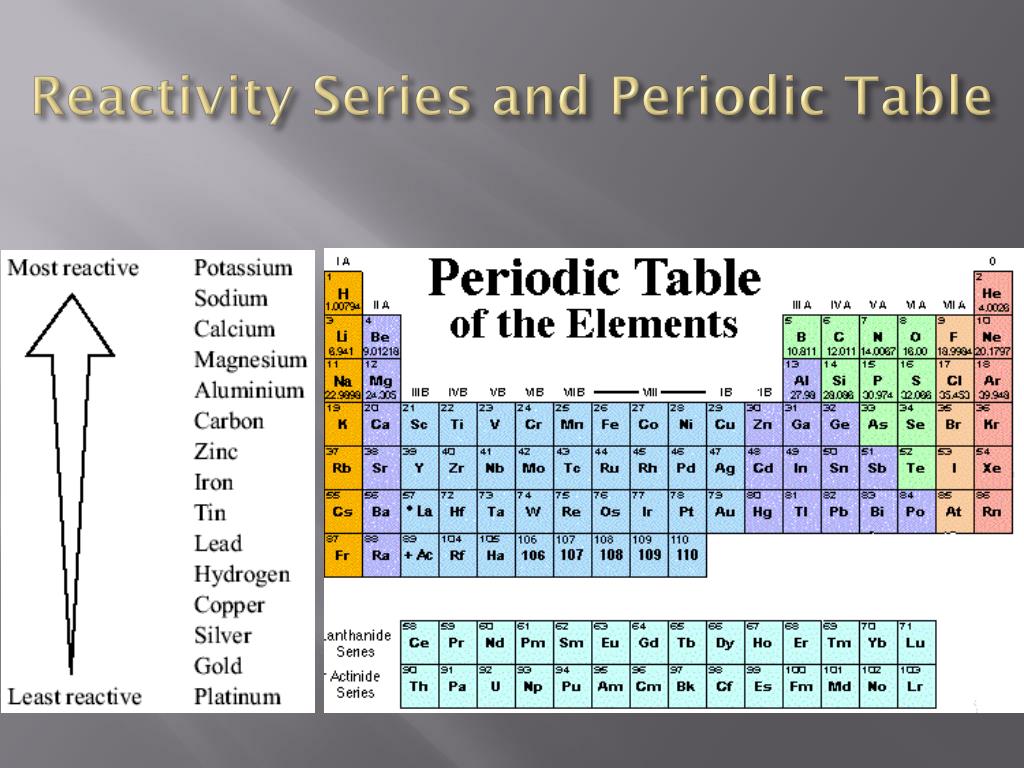
The affinity of electrons – refers to the energy that is released when an electron is gained by an atom. On moving in a diagonal direction, atomic radius tends to increase. On moving from left to right, it increases, and on moving from top to bottom, it increases. Ionisation energy increases on moving horizontally across a period, whereas on moving in the downward direction, the ionisation energy decreases.Ītomic radii – the distance between the outermost electron and the nucleus is known as the atomic radius. Ionisation energy – this energy helps the atoms to remove the electrons. An exception in group 3 elements is present where electronegativity increases towards thallium. On moving in the downward direction, the electronegativity decreases. On moving from left to right horizontally in a periodic table, there is an increase in electronegativity. The Pauling scale is used to measure electronegativity. Some periodic trends and chemical reactivity in the periodic table are as follows –Įlectronegativity – measures the ability of a molecule to gain electrons within a chemical bond. The trends of the elements in a periodic table depend on various factors like electronegativity, ionisation energy, electron affinity, atomic radii, metallic character, and chemical reactivity. Periodic trends refer to the patterns of the properties of the chemical elements. The periodic trends and chemical reactivities of elements are shared below. The gas elements do not gain or lose their electrons because the electronic configuration is filled. Hence the elements on the left side in the downwards direction are the highly reactive elements. In a period, the chemical reactivity decreases on moving towards the right, whereas in a group, chemical reactivity increases on moving downwards. Hence the electronegativity and the ionisation energy determine a chemical reaction. The higher the reactivity of the element, the easier it is to combine. The electrons between the elements are shared, or the elements lose or gain the electrons. 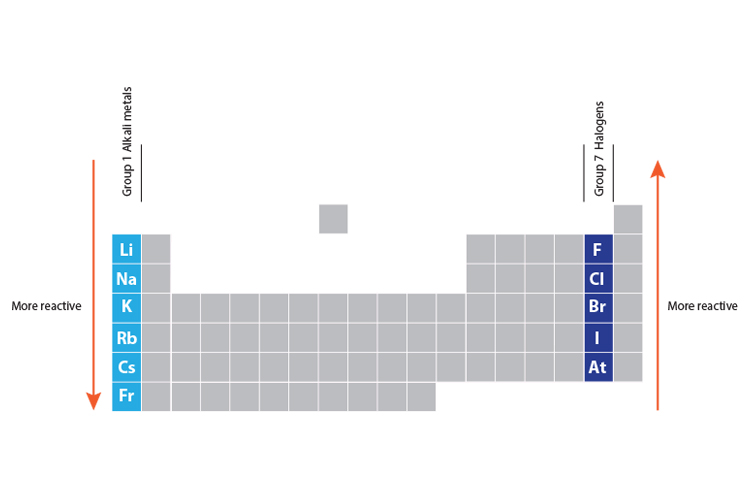
If it's wrong please correct me.The rate at which a chemical substance or an element undergoes a chemical reaction is known as reactivity. I am not sure of how correct my knowledge is. This is the way I personally see it at least. So they would still be more active going to the left-hand side of the table. Second, assuming they could do both (lose and gain) it would still be far easier to lose 2 or 3 electrons than add like 6 more to fill the valance shell. Now intuitively it could be thought that an atom would like to have an octet of valance electrons and since most of the nonmetals are on the right-hand side of the table, the closest "octet" would be to the right, achieved by gaining electrons, and the closer it is to the right, the more it wants that electron (becomes more active like halogens).īut for metals it's the very opposite, they want to LOSE electrons because Nonmetals can do both, lose and gain electrons. Well first off metals can ONLY lose electrons, so they will always either gain a positive charge or will have a positive oxidation state. A screenshot is preferable to a picture of your laptop screen. Please do not ask for help acquiring, preparing, or handling illicit substances or for help with any activity that does not fall within the confines of whatever laws apply to your particular location.īonus points: If submitting a picture please make sure that it is clear. Any infractions will be met with a temporary ban at the first instance and a permanent ban if there is another. It is also important that you describe the specific part of the problem you are struggling with. It is OK if you are a little (or a lot!) stuck, we just want to see that you have made an effort. 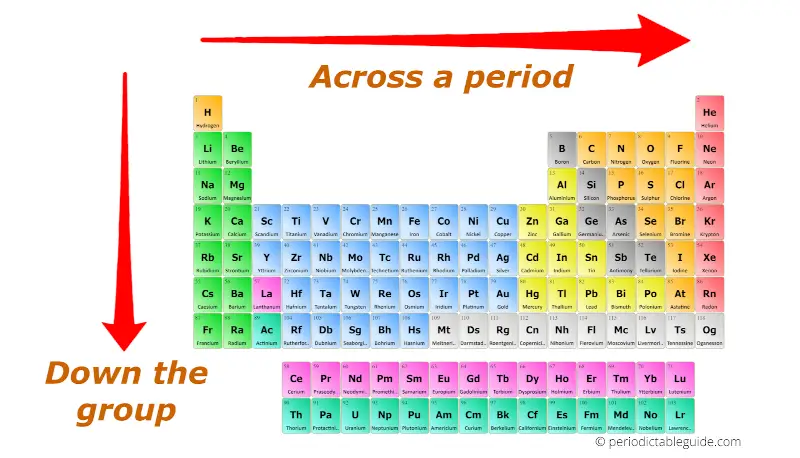
Please complete any questions as much as you can before posting. We will not do your homework for you, so don't ask. Please flair yourself and read over the rules below before posting. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
